Which Element Forms An Ion Larger Than Its Atom
Which Element Forms An Ion Larger Than Its Atom - Web the element that forms an ion larger than its atom is chlorine (cl). Web an ion's size, compared to its parent atom, depends on whether it gains or loses electrons. Because most elements form either a cation or an. For ions of the same charge (e.g. Web the ionic radii of cations and anions are always smaller or larger, respectively, than the parent atom due to. A) lead b) sulfur c) oxygen d) fluorine Anions are therefore larger than the parent atom. This can only be possible if the ion is an anion, and anions. Web which element forms an ion that is smaller than its atom? Web as mentioned, we are looking for an ion larger than its initial atom.
PPT Ions PowerPoint Presentation, free download ID6738771
Web an ion's size, compared to its parent atom, depends on whether it gains or loses electrons. This can only be possible if the ion is an anion, and anions. Web which element forms an ion that is smaller than its atom? Web the ionic radii of cations and anions are always smaller or larger, respectively, than the parent atom.
PPT Chapter 7 Periodic Properties of the Elements PowerPoint Presentation ID1800688
Web as mentioned, we are looking for an ion larger than its initial atom. Web cations are always smaller than the neutral atom and anions are always larger. For ions of the same charge (e.g. Web the ionic radii of cations and anions are always smaller or larger, respectively, than the parent atom due to. This happens due to the.
7.3 Sizes of Atoms and Ions Chemistry LibreTexts
Anions are therefore larger than the parent atom. Web cations are always smaller than the neutral atom and anions are always larger. Web as mentioned, we are looking for an ion larger than its initial atom. Web which element forms an ion that is smaller than its atom? A) lead b) sulfur c) oxygen d) fluorine
CH150 Chapter 3 Ions and Ionic Compounds Chemistry
In the same group) the size increases as we go down a group in the periodic table. Web which element forms an ion that is smaller than its atom? Web the ionic radii of cations and anions are always smaller or larger, respectively, than the parent atom due to. Web as mentioned, we are looking for an ion larger than.
Periodic Table of Electronegativities
Web an ion's size, compared to its parent atom, depends on whether it gains or loses electrons. Web as mentioned, we are looking for an ion larger than its initial atom. As the principle quantum increases the size of both the parent atom and the ion will increase. Web cations are always smaller than the neutral atom and anions are.
Classification of Elements & Periodicity in Properties askIITians
Web which element forms an ion that is smaller than its atom? In the same group) the size increases as we go down a group in the periodic table. As the principle quantum increases the size of both the parent atom and the ion will increase. This can only be possible if the ion is an anion, and anions. Web.
CH150 Chapter 3 Ions and Ionic Compounds Chemistry
A) lead b) sulfur c) oxygen d) fluorine This happens due to the process of acquiring. For ions of the same charge (e.g. Web which element forms an ion that is smaller than its atom? Because most elements form either a cation or an.
PPT Noble Gases and Valence e Ionization Energy and Bonding PowerPoint Presentation ID5875405
Web the element that forms an ion larger than its atom is chlorine (cl). A) lead b) sulfur c) oxygen d) fluorine Web cations are always smaller than the neutral atom and anions are always larger. Web the ionic radii of cations and anions are always smaller or larger, respectively, than the parent atom due to. Because most elements form.
Atomic Radius and Ionic Radius
Because most elements form either a cation or an. Web which element forms an ion that is smaller than its atom? Web the ionic radii of cations and anions are always smaller or larger, respectively, than the parent atom due to. This happens due to the process of acquiring. Web cations are always smaller than the neutral atom and anions.
3.1 Ionic Atoms Chemistry LibreTexts
Web which element forms an ion that is smaller than its atom? Because most elements form either a cation or an. In the same group) the size increases as we go down a group in the periodic table. For ions of the same charge (e.g. A) lead b) sulfur c) oxygen d) fluorine
Web the element that forms an ion larger than its atom is chlorine (cl). As the principle quantum increases the size of both the parent atom and the ion will increase. A) lead b) sulfur c) oxygen d) fluorine In the same group) the size increases as we go down a group in the periodic table. Anions are therefore larger than the parent atom. Web as mentioned, we are looking for an ion larger than its initial atom. Web which element forms an ion that is smaller than its atom? Because most elements form either a cation or an. This happens due to the process of acquiring. For ions of the same charge (e.g. Web an ion's size, compared to its parent atom, depends on whether it gains or loses electrons. Web the ionic radii of cations and anions are always smaller or larger, respectively, than the parent atom due to. This can only be possible if the ion is an anion, and anions. Web cations are always smaller than the neutral atom and anions are always larger. Web which element forms an ion that is smaller than its atom?
This Happens Due To The Process Of Acquiring.
Anions are therefore larger than the parent atom. In the same group) the size increases as we go down a group in the periodic table. Web as mentioned, we are looking for an ion larger than its initial atom. Web which element forms an ion that is smaller than its atom?
Web Cations Are Always Smaller Than The Neutral Atom And Anions Are Always Larger.
Web the ionic radii of cations and anions are always smaller or larger, respectively, than the parent atom due to. As the principle quantum increases the size of both the parent atom and the ion will increase. For ions of the same charge (e.g. Web an ion's size, compared to its parent atom, depends on whether it gains or loses electrons.
Because Most Elements Form Either A Cation Or An.
This can only be possible if the ion is an anion, and anions. A) lead b) sulfur c) oxygen d) fluorine Web the element that forms an ion larger than its atom is chlorine (cl). Web which element forms an ion that is smaller than its atom?




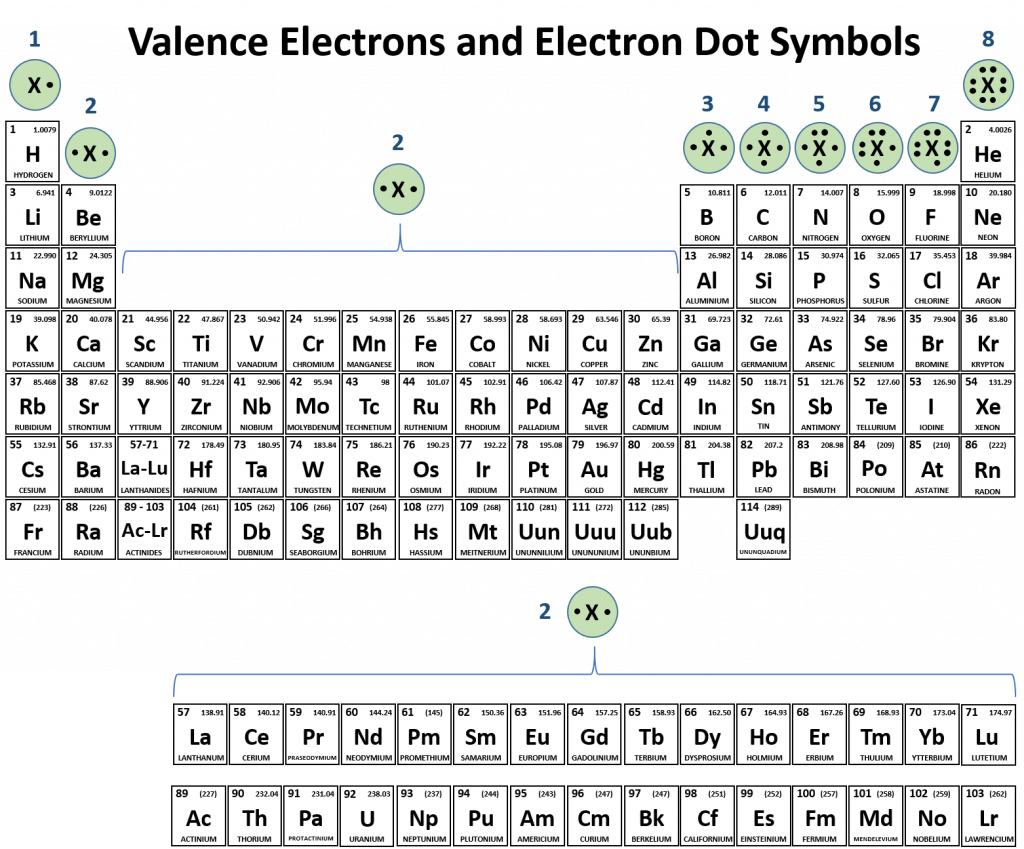
.PNG)




